














| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |
Tilmicosin Injection 30% 100ml vial
Contains per ml:
Tilmicosin (as phosphate)...............300 mg.
Solvents ad ........................1 ml.
For first-line control and treatment of Bovine Respiratory Disease (BRD). Also used for the treatment of respiratory bacterial diseases in large ruminants, caused by Pasteurella miltocida, Mannheimia (Pasteurella) haemolytica, infection processes associated with Haemophilus somnus, Histophilus spp. etc.
For subcutaneous administration:
Cattle (Pneumonia): 1 ml per 30 kg body weight (10 mg/kg).
Cattle (Interdigital necrobacillosis): 0.5 ml per 30 kg body weight (5 mg/kg).
Sheep (Pneumonia and mastitis): 1 ml per 30 kg body weight (10 mg/kg).
Sheep (Footrot): 0.5 ml per 30 kg body weight (5 mg/kg).
For meat:
Cattle: 60 days. | Sheep: 42 days.
For milk:
Sheep: 15 days.
Shelf-life: 2 years
Storage: Tightly sealed, protect from light, store below 25ºC.
CONSULT YOUR VETERINARIAN BEFORE USING
If you need it, we can customize exclusive formulas, packaging, and everything you want for you. If you need a product manual, please contact us. Thank you.
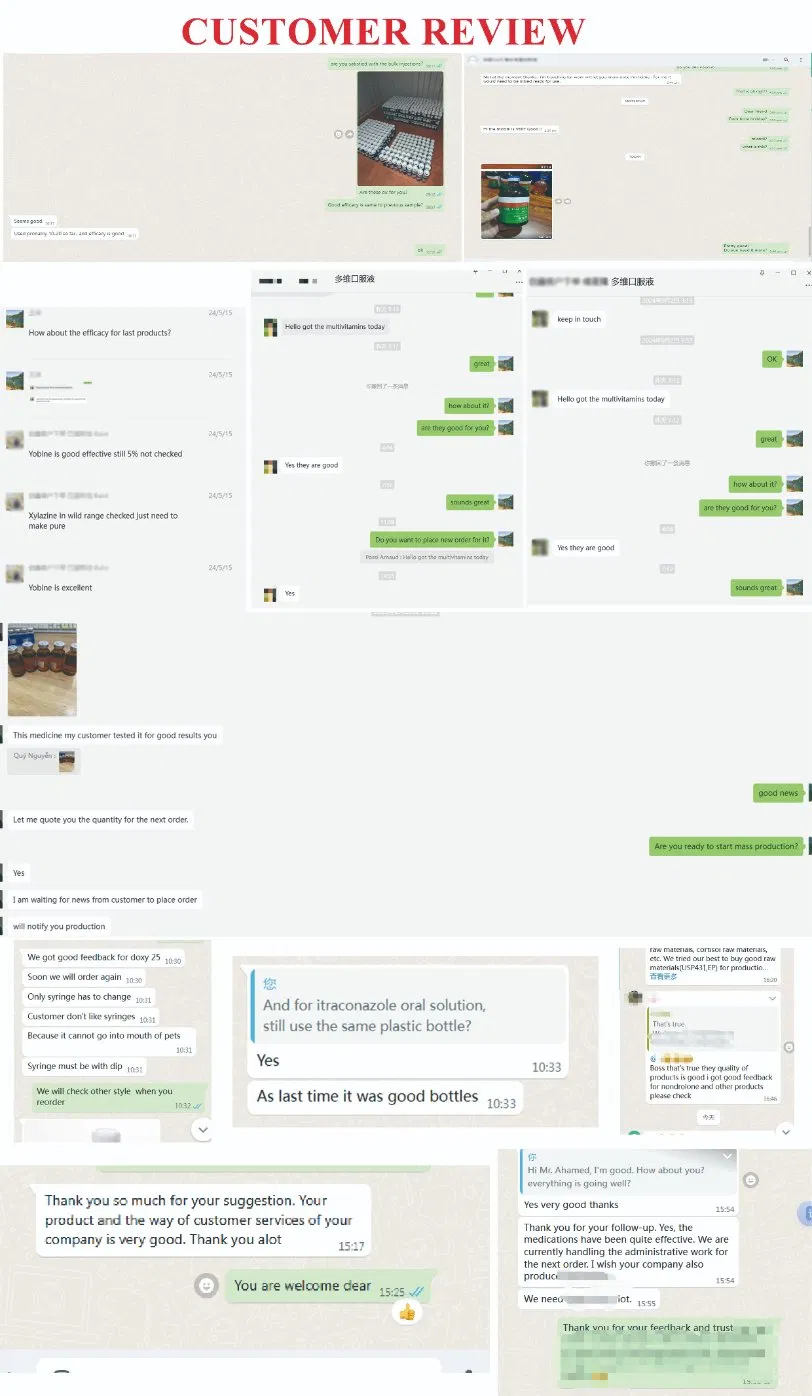







 Tucson Medical
Tucson Medical